|
ramé-hart instrument co. March 2014 Newsletter |
| Surface Tension and Viscosity |
|
Here's a question that I've been asked more than once: What's the relationship between surface tension and viscosity? Intuition tells us that a thick liquid with high viscosity will have a higher surface tension than a thin liquid with low viscosity. However, this may not be a safe conclusion. Viscosity is the measurement of internal resistance to change in shape by the forces of shear or tensile stress in a fluid in motion. It explains the friction between molecules which can change based on pressure and velocity. Viscosity can get involved. For example, some fluids conform to Newton's law, such as water, and are considered Newtonian fluids. While other fluids, such as ketchup, are considered non-Newtonian because their viscosity is dependant on shear rate. Have you ever noticed that ketchup doesn't want to budge until you whack the bottle - and then once it does starts flowing, it doesn't want to stop? We don't really get involved in measuring viscosity but let us say that it's a dynamic measurement which is about capturing properties of fluids in motion. Surface tension, by contrast, is actually a much simpler concept. It's characterized by what appears to be an elastic sheet on a liquid's surface. Surface tension is what allows a water strider to walk on water.
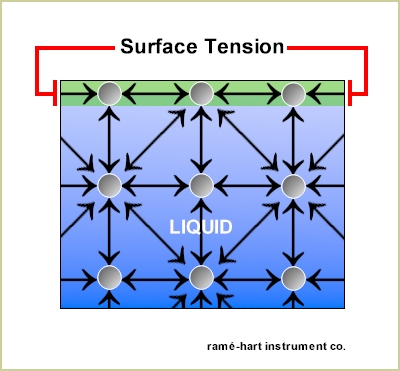
The water molecules inside the liquid (shown in blue in the graphic below) are attracted to one another through cohesive forces which balance out to zero. The (green) molecules at the liquid's surface, however, do not have as many molecules to attract to and are thus pulled inward causing the surface of a liquid to behave like an elastic sheet. Surface tension is what causes drops to bead up on a surface. And it's the balance between the surface tension at the gas/liquid interface and the surface energy at the liquid/solid interface that drive the contact angle.
Surface tension is one thing that we are very good at measuring using ramé-hart instruments. However, surface tension, is generally a static measurement. We often measure it using a pendant drop. Viscosity, on the other hand, is about movement and velocities. So, while it's tempting to draw a correlation between surface tension and viscosity, we conclude that there is none. Viscosity has to do with the property of fluids in motion; surface tension describes the behavior of the outermost layer of molecules of a liquid at the liquid/gas interface. There is no correlation. Take honey, for instance, which has a
very high viscosity: its surface tension is actually lower than that of
water which has much lower viscosity. Acetone, by contrast, has a lower
viscosity than water but its surface tension is also lower.
And so it is between any pair of liquids - there is no rule of thumb or
correlation between surface tension and viscosity. Nonetheless, despite
there there being no relationship between these two metrics, it is
generally true that as temperature increases, both viscosity and surface
tension decrease. |
| Viscosity and Dispensing |
|
While we conclude that there is no
correlation between viscosity and surface tension, we cannot ignore
viscosity - especially when dispensing liquids. For that reason we offer
a wide range of dispensing needles which can be used with our manual
microsyringe, our
Automated Dispensing System, and
Elevated Temperature Syringe. For water we use a 22 gauge needle.
For liquids with higher viscosity we use a lower gauge needle: 18 gauge
for glycerol, 12 gauge for melted polymers (using our Elevated Temperature
Syringe). For thin low viscosity liquids we use higher gauge needles -
26 gauge for hexane, 30 gauge for acetone, for example. We keep in stock
dispensing needles from 8 gauge to 33 gauge although 12 gauge to 30
gauge are the most common with 22 gauge the standard.
For a complete chart of our needles including inside and outside diameters, see this chart: http://www.ramehart.com/pdf/needles.pdf. To order straight dispensing needles, go to: http://www.ramehart.us/straight-304-stainless-steel-reusable-dispensing-needle/ and for inverted needles, go to: http://www.ramehart.us/inverted-304-ss-needle/. ramé-hart is also the world's number one fabricator of coaxial and triaxial needles; get started here: http://www.ramehart.com/coaxial_needles.htm |
|
Regards,
Carl Clegg |