|
|
| Contact Angle Spreading Coefficient |
|
Generally, when a drop is deposited on a surface and it spreads out completely we say that wetting is complete. This is the result of high surface energy. The contact angle is approaching zero but is not measurable using conventional goniometrical methods. We tend to categorize all such surfaces as having the same wetting property: complete wetting. However, even when wetting appears to be complete, there may be a variety of surfaces and surface properties which can be discerned through more careful examination. The first question is whether complete wetting is the result of absorption or simply a high energy surface or some combination. The easiest way to determine this is to perform a captive bubble contact angle measurement. The surface is immersed in a liquid (water, e.g.) and an air bubble is formed on the underside of the surface. If the solid is porous or semi-porous, it will become saturated and the contact angle can then be measured. Two different surfaces, for example, which both exhibit complete wetting using a sessile drop in an air phase may exhibit varying contact angles when measured using the captive bubble method thus providing a way to differentiate between the two surfaces when they are fully hydrated.
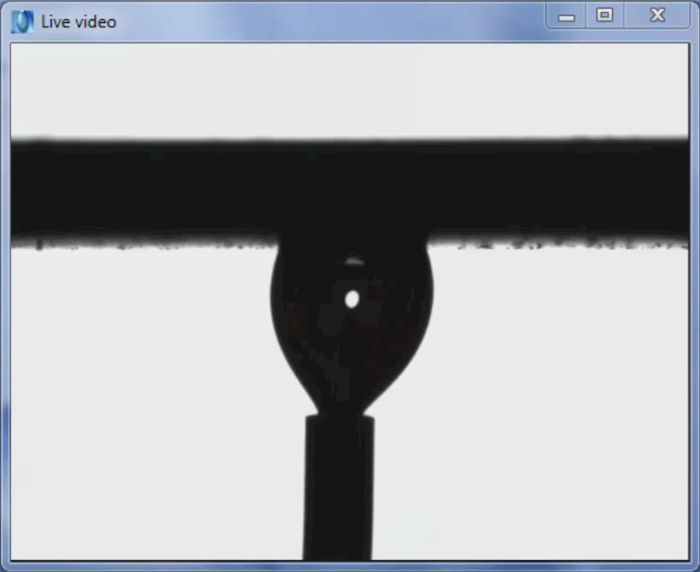
The video above shows a captive bubble measurement using DROPimage Advanced. Engineered tissue, breast implants, contact lenses, disposable diapers and other products fabricated from hydrogels and superabsorbent polymers benefit from the captive bubble method as the traditional method using a sessile drop in a gaseous external phase results in fast absorption which makes it hard to measure.
Another condition that results in complete wetting is when there is no absorption but the solid simply exhibits high surface energy - that is, the solid surface energy is greater than the liquid's surface tension and the solid/liquid interfacial free energy combined. These energies are explained by Young's Equation. See the graphic above. The following equation is used to calculate the spreading coefficient (S): S = gsv - (gsl + glv) When S > 0, complete wetting occurs. When S < 0, then partial wetting occurs and the contact angle is greater than 0°. Thus, an arrangement that leads to complete wetting will include a high surface energy solid (e.g., glass or silicon) with a low surface tension liquid (e.g., acetone, heptane). The spreading coefficient S is used to build the Young-Dupré equation: S = glv (cosθ - 1) Using the Young-Dupré equation, we determine that the equation cannot be solved if contact angle is 0°. Thus the Young-Dupré equation is only solvable for partial wetting conditions - that is, when S < 0. When contact angle measurements are used to derive solid surface free energy, it's helpful that the contact angle is greater than 0°, that is, S < 0. This also explains why pure deionized water is the most recommended test liquid for contact angle. Not only is it cheap and plentiful but very few liquids have a higher surface tension. (Mercury does but it's not a safe or practical liquid to work with.) There are times when wetting appears complete but is not. If you can look above the sessile drop and see a circle defining the three-phase line, then it's possible to measure the contact angle even if it's not possible to measure an extremely low contact angle using the traditional goniometric method from the profile view. In order to successfully measure the contact angle when it's below 5°, use our proprietary Optical Overhead Imaging Kit which is available for use with any instrument that has DROPimage Advanced. This novel method captures the precise surface area of the drop on the surface from the top view and then mathematically calculates the contact angle. For more details on this product, including a PDF brochure and pictures, go to: http://www.ramehart.com/accessories.htm#100-31. In conclusion, there are tools available such as the captive bubble method and the ramé-hart Optical Overhead Imaging Kit which can increase our understanding of wetting behavior on high-energy surfaces when wetting is complete and not measurable using traditional contact angle methods.
|
| Happy New Year |
| From all of us at ramé-hart instrument co., we wish you a happy and prosperous New Year. We look forward to working with you in 2016 on all of your surface science instrumentation needs. |
|
Regards,
Carl Clegg |